The Core Biological Principle
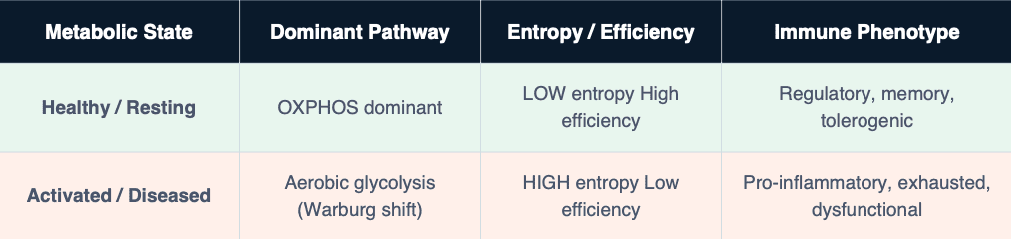
Every macrophage makes a thermodynamic choice. It can run on Oxidative Phosphorylation (OXPHOS) — a high-efficiency, low-entropy metabolic state — or switch to aerobic glycolysis — a fast, wasteful, high-entropy mode. This thermodynamic axis determines whether the immune cell heals or damages the host.
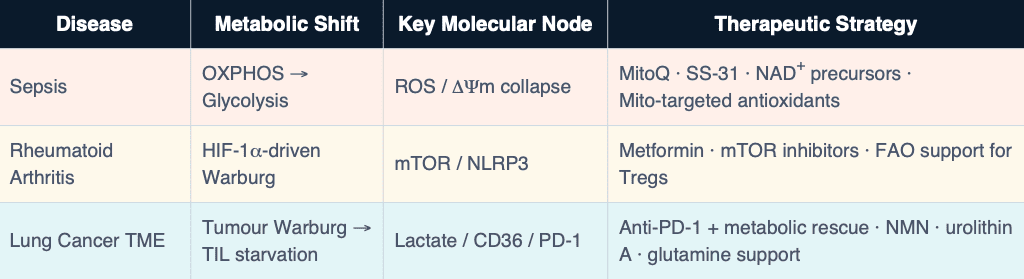
This bioenergetic trade-off is not a side effect of disease. It is the molecular engine of disease. The same logic plays out identically across three clinically distinct conditions: sepsis, rheumatoid arthritis, and lung cancer.
The Bioenergetic Phenotype Comparison
1. Sepsis — When the Engine Overheats
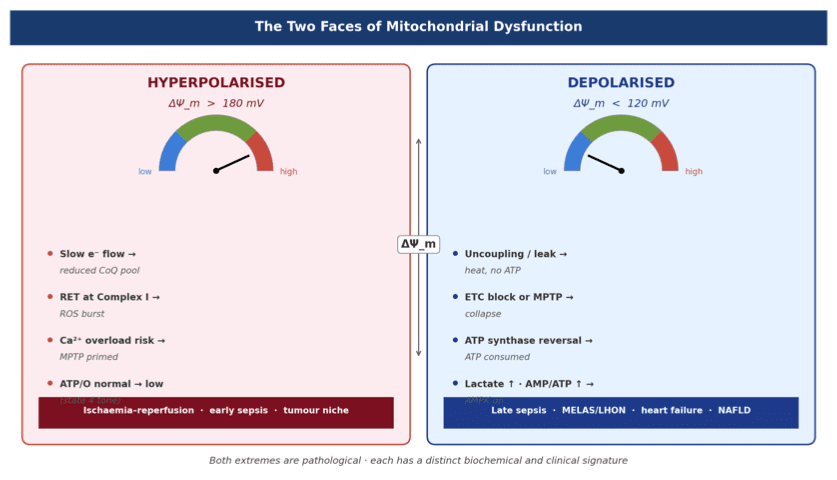
In sepsis, bacterial signals collapse the mitochondrial membrane potential (∆Ψm), flood the cell with reactive oxygen species (ROS), and force a metabolic switch from OXPHOS to glycolysis. The result is M1 macrophage hyperpolarisation: massive IL-1β, TNF-α, and IL-6 release — the cytokine storm. This is not just inflammation; it is thermodynamic failure. The cell dissipates energy as heat and entropy instead of extracting useful biological work.
Molecular Mechanisms
• ROS burst from Complex I/III → HIF-1α stabilisation → glycolytic gene upregulation
• Loss of ∆Ψm → impaired ATP synthase coupling → proton leak as heat
• mtDNA released into cytosol → cGAS-STING activation → amplified innate immune response
• Succinate accumulation → HIF-1α and IL-1β post-translational stabilisation
✓ Clinical Translation +
• NAD+ precursors (NMN, NR): restore Complex I cofactor availability, rescue OXPHOS capacity, and reduce M1 cytokine production in preclinical sepsis models.
• Mitochondria-targeted antioxidants (MitoQ, SS-31): scavenge IMM-generated superoxide, preserve ∆Ψm, and attenuate organ injury in CLP models.
• Biomarker opportunity: OCR/ECAR ratio (oxygen consumption vs. extracellular acidification) as a bedside metabolic index of macrophage inflammatory state.
2. Rheumatoid Arthritis — Entropy as a Chronic Tenant
The RA synovial joint is a permanently high-entropy microenvironment: hypoxic, glucose-depleted, acidic, and flooded with lactate. CD4+ T cells, macrophages, and fibroblast-like synoviocytes converge on aerobic glycolysis to survive. This metabolic shift is the inflammation — it sustains HIF-1α, fuels NF-κB, and keeps the NLRP3 inflammasome active. Meanwhile, Tregs (which depend on fatty acid oxidation and OXPHOS for their suppressive function) are metabolically starved in the same environment.
Molecular Mechanisms
• HIF-1α → GLUT1/LDHA upregulation → increased glycolytic flux → IL-17, IL-6 production
• Mitochondrial ROS → NF-κB and NLRP3 activation → sustained cytokine loop
• mTORC1 activation → suppresses OXPHOS → biases T cells away from Treg toward Th17 fate
• FAO-dependent Tregs: thermodynamic substrate starvation (low lipid availability in hypoxic joint) = loss of immune regulation
✓ Clinical Translation +
• Metformin: Complex I inhibitor → activates AMPK, restores metabolic balance, reduces glycolytic flux in RA immune cells. Anti-inflammatory effect independent of its glucose-lowering action.
• mTOR inhibitors (rapamycin analogues): shift T cell fate from Th17 toward Treg by derepressing OXPHOS and FAO.
• Biomarker opportunity: P/O ratio (ATP per O2 consumed) and NAD+ /NADH in synovial immune cells as quantitative disease activity metrics.
3. Lung Cancer — The Tumor as a Thermodynamic Trap
Lung tumours export entropy into their microenvironment. They consume glucose ferociously via the Warburg effect, release lactate that acidifies the milieu, and deplete O2 — creating a thermodynamic prison for tumour-infiltrating lymphocytes (TILs). TILs show declining oxygen consumption rates, reduced mitochondrial mass, and collapsed ∆Ψm. T cell exhaustion is not merely a transcriptional programme — it is a bioenergetic collapse. The tumour does not just hide from immunity. It starves it.
Molecular Mechanisms
• Warburg glycolysis in tumour cells → lactate efflux → TME acidification → T cell OXPHOS inhibition
• Glucose/glutamine depletion in TME → substrate-starved TILs → loss of effector ∆G
• Hypoxia → HIF-1α in tumour → VEGF/PD-L1 upregulation → immune exclusion and checkpoint exhaustion
• CD36high TIL phenotype: excess lipid uptake → mitochondrial lipid overload → ferroptosis susceptibility
✓ Clinical Translation +
• Metabolic + checkpoint combination: OXPHOS enhancers (urolithin A, NMN) + anti-PD-1 show synergistic TIL reinvigoration in preclinical NSCLC models.
• Nutritional intervention: methionine restriction reduces Treg suppression; glutamine supplementation restores TIL respiratory capacity.
• Biomarker opportunity: Seahorse OCR profiling of TILs pre-therapy; hyperpolarised 13C-pyruvate MRI for real-time TME metabolic mapping.
Thermodynamic & Quantitative Perspective
Each disease represents a measurable deviation from thermodynamic efficiency. The key variables are not just molecular — they are quantifiable bioenergetic parameters that can be tracked clinically:
• ∆Ψm (mitochondrial membrane potential): Index of proton motive force and OXPHOS coupling. Collapse precedes immune dysfunction.
• P/O ratio (phosphorylation efficiency): ATP produced per O■ consumed. Declines with inflammatory reprogramming.
• OCR/ECAR ratio: Oxygen consumption vs. extracellular acidification rate — a real-time OXPHOS/glycolysis balance index.
• NAD+ /NADH ratio: Master redox sensor. Low NAD+ = impaired OXPHOS, increased entropy, impaired sirtuin-mediated immune regulation.
• mtDNA copy number / cytosolic mtDNA: Proxy for mitochondrial entropy accumulation and DAMP-mediated innate activation.
Unified Clinical Framework
Where This Is Going: Next-Generation Immune Biomarkers
The next generation of immune biomarkers will not be limited to cytokines alone. They will be metabolic:
• OCR/ECAR ratio in patient peripheral blood immune cells
• Mitochondrial membrane potential (∆Ψm) as an immune activation index
• NAD+ /NADH ratio as a systemic bioenergetic health score
• mtDNA release rate as a real-time entropy production marker
• Urinary lactate/pyruvate ratio as a non-invasive glycolytic shift readout
And the therapies that follow will not just block receptors — they will restore thermodynamic order to immune cells that have lost it. This is the promise of immunometabolism: not more immunosuppressants, but smarter bioenergetic strategies that give immune cells back what disease took from them.
Selected Evidence Base
1. Ji F et al. Crosstalk of mitochondrial dysfunction and macrophage polarization in sepsis. Front Immunol. 2026.
2. Xie R et al. Roles of immune cell metabolism in rheumatoid arthritis. Front Immunol. 2026.
3. Eivazzadeh Y et al. Immunometabolism in lung cancer. iScience. 2026.
4. Dwivedi V et al. Mitochondrial dysfunction and cellular senescence in ageing sarcopenia. Mol Biol Rep. 2026.
5. Seledtsov V. Mitochondria-targeted therapy in anti-aging medicine. J Biol Methods. 2026.
6. Glogowski PA et al. Reprogramming the mitochondrion in atherosclerosis. Antioxidants. 2025.
7. Xu Y et al. Nutritional intervention alleviates T cell exhaustion and empowers anti-tumor immunity. Front Immunol. 2026.
8. Zhang XY et al. The role of CD36 in immune function. Front Immunol. 2026.
This document is an evidence-based scientific communication intended for medical professionals, PhD researchers, and advanced students. All claims reflect peer-reviewed literature as of Q1 2026. Not intended as direct clinical guidance.