About this course booklet
Complex I is the first and largest enzyme of the mitochondrial respiratory chain, where electron free energy is converted into proton translocation across the inner mitochondrial membrane (IMM).
The energy that moves a proton across the IMM is not chemical in the ATP sense — it is electron free energy, harvested from NADH as electrons flow down a redox gradient. The global driving force is the difference in midpoint potential between NADH/NAD⁺ (≈ −320 mV) and O₂/H₂O (+820 mV), corresponding to ΔG°’ ≈ −220 kJ·mol⁻¹ per 2 e⁻.
Complex I performs the first conversion step of this energy into the proton motive force (Δp), capturing part of this free energy while the remainder is dissipated as heat and entropy production.
This educational eBook (April 2026) is designed for PhD and MD students and provides a rigorous framework linking electron transfer, molecular mechanisms, and thermodynamics to the physical act of pumping protons.
About the booklet structure
This 10-page PDF is structured as a compact course:
- Cover
- Preface
- Table of contents
- 2 core chapters
- 2 appendices (equations and glossary)
Core concepts developed in the booklet
Electron free energy as the driving force
- ΔG°’ = −nFΔE°’ as the fundamental relation
- NADH → O₂ releases ~−220 kJ·mol⁻¹
- Complex I captures the first ~−70 kJ·mol⁻¹ step
- Proton pumping as thermodynamically uphill work
Molecular mechanism — a redox-driven piston
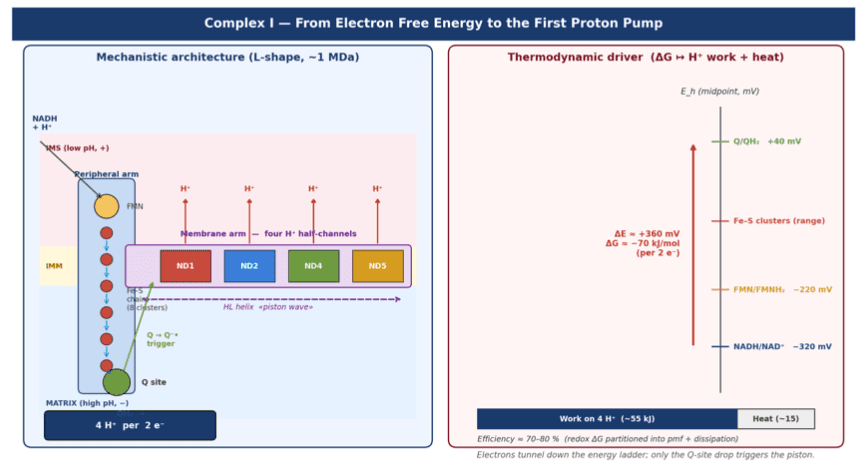
- L-shaped structure (~1 MDa, 45 subunits)
- Redox arm: FMN + Fe–S clusters (N1a → N2)
- Membrane arm: proton channels in ND1, ND2, ND4, ND5
- Semiquinone (Q⁻•) as the mechanical trigger
- HL helix as a long-range conformational transducer (~60 Å)
Electron transfer pathway:
NADH → FMN → Fe–S clusters → Q → QH₂
The redox drop does not directly bind protons — it triggers a conformational wave that drives proton translocation via alternating-access channels.
Stoichiometry and system behavior
- 1 NADH → 2 e⁻ → 4 H⁺ pumped
- Total respiratory yield: 10 H⁺ per NADH
- Reverse electron transport (RET) under high Δp
- Complex I as a major ROS source in hyperpolarised states
- A↔D transition regulating activity during ischemia
Quantitative and thermodynamic framework
- ΔE (NADH → Q) ≈ +0.36 V
- ΔG ≈ −70 kJ·mol⁻¹
- Δp ≈ 180–220 mV
- Work required: ~+77 kJ·mol⁻¹ for 4 H⁺
- Efficiency ≈ 70–80%
Key relations:
- ΔG°’ = −nFΔE°’
- Δp = ΔΨ_m − (2.303 RT/F)·ΔpH
Complex I operates close to equilibrium but deliberately dissipates part of the energy to maintain forward flux — an expression of the second law of thermodynamics.
Clinical and translational relevance
- Complex I deficiencies (Leigh syndrome, MELAS, LHON)
- Ischemia-reperfusion injury and RET-driven ROS
- Metformin as a reversible Complex I inhibitor
- Hyperpolarisation vs depolarisation pathologies
- Central role in mitochondrial dysfunction and energy failure
Key learning anchors
- “The first proton is paid for by ~70 kJ·mol⁻¹ of electron free energy”
- Semiquinone (Q⁻•) is a mechanical trigger, not an energy store
- HL helix = long-range mechanical transducer
- Efficiency < 100% = necessary entropy production
Who is this for?
- PhD students in bioenergetics and molecular biology
- Medical doctors interested in mitochondrial physiology
- Researchers in redox biology
- Clinicians exploring mitochondrial dysfunction
Key insight
Complex I is a redox-driven mechanical engine that converts electron free energy into proton pumping, initiating the proton motive force and cellular energy production.
Note: The full PDF is in English.